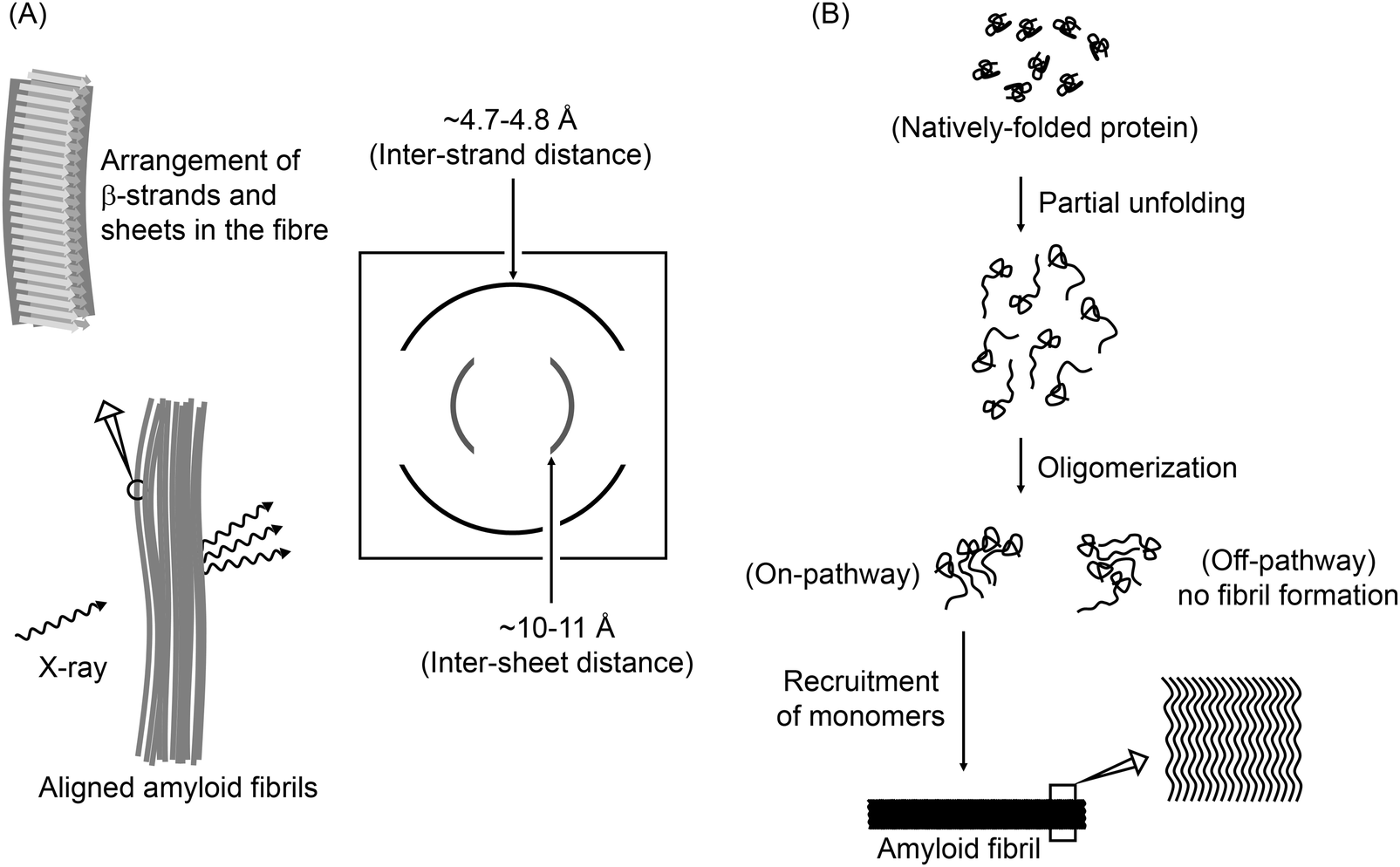
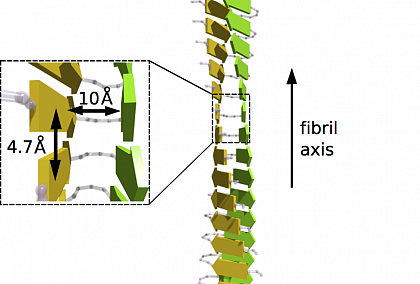
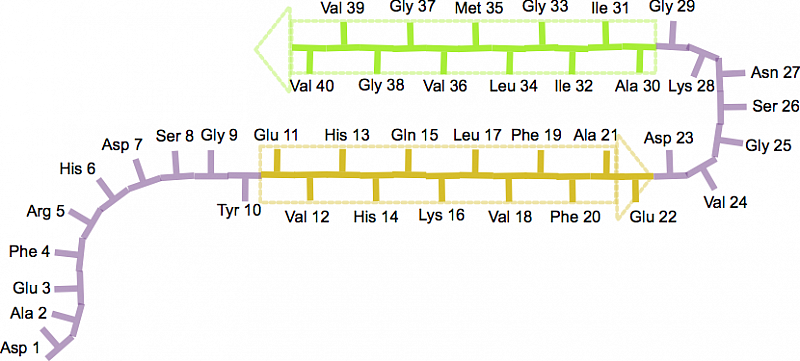
Identification of two principal amyloid-driving segments in variable domains of Ig light chains in systemic light-chain amyloidosis - Journal of Biological Chemistry

Some structural models of amyloid fibrils. a) view down the fibre axis,... | Download Scientific Diagram
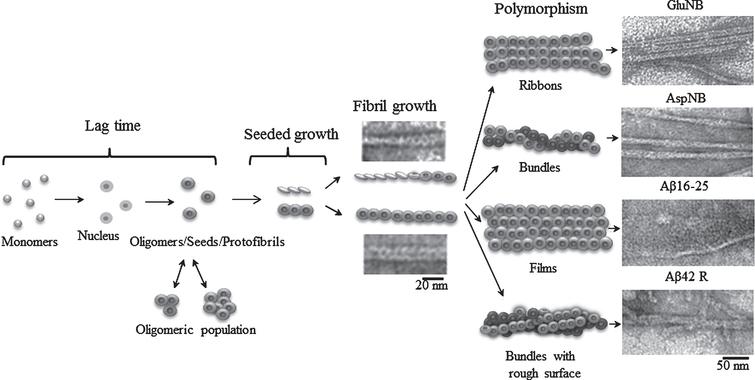
Rosetta Stone for Amyloid Fibrils: The Key Role of Ring-Like Oligomers in Amyloidogenesis - IOS Press

Rapid Fluorescence Lifetime Imaging Reveals That TRPV4 Channels Promote Dysregulation of Neuronal Na+ in Ischemia | Journal of Neuroscience

Physical properties of the cytoplasm modulate the rates of microtubule polymerization and depolymerization - ScienceDirect

Peptide αA66-80 forms amyloid-like fibrils. The peptide αA66-80 was... | Download Scientific Diagram
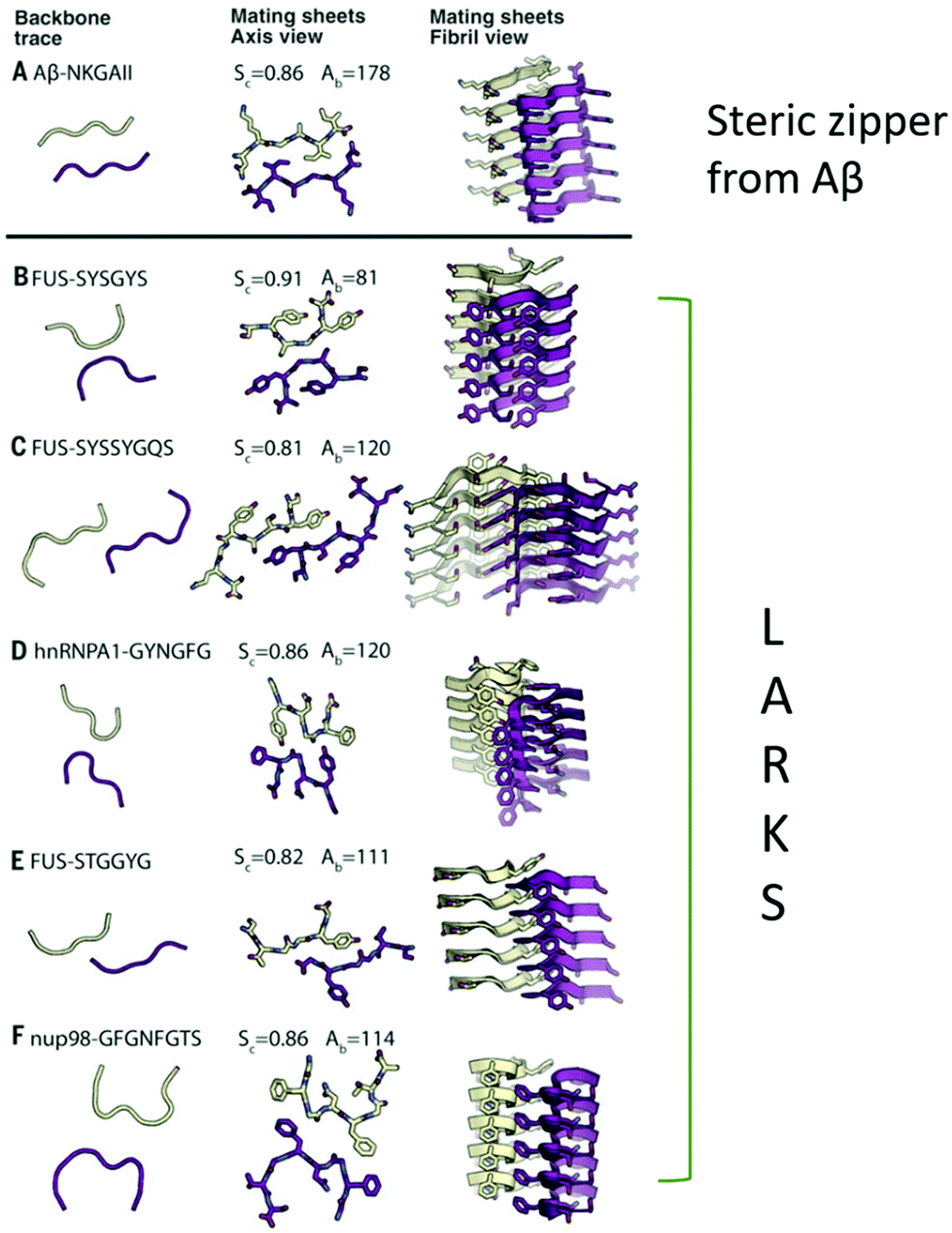
Half a century of amyloids: past, present and future - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C9CS00199A

Physical properties of the cytoplasm modulate the rates of microtubule polymerization and depolymerization - ScienceDirect

Nicking and fragmentation are responsible for α‐lactalbumin amyloid fibril formation at acidic pH and elevated temperature - Rahamtullah - 2021 - Protein Science - Wiley Online Library
Structural and functional diversity among amyloid proteins: Agents of disease, building blocks of biology, and implications for

Computational Design of Functional Amyloid Materials with Cesium Binding, Deposition, and Capture Properties | The Journal of Physical Chemistry B

Mechanism of Islet Amyloid Polypeptide Fibrillation at Lipid Interfaces Studied by Infrared Reflection Absorption Spectroscopy - ScienceDirect